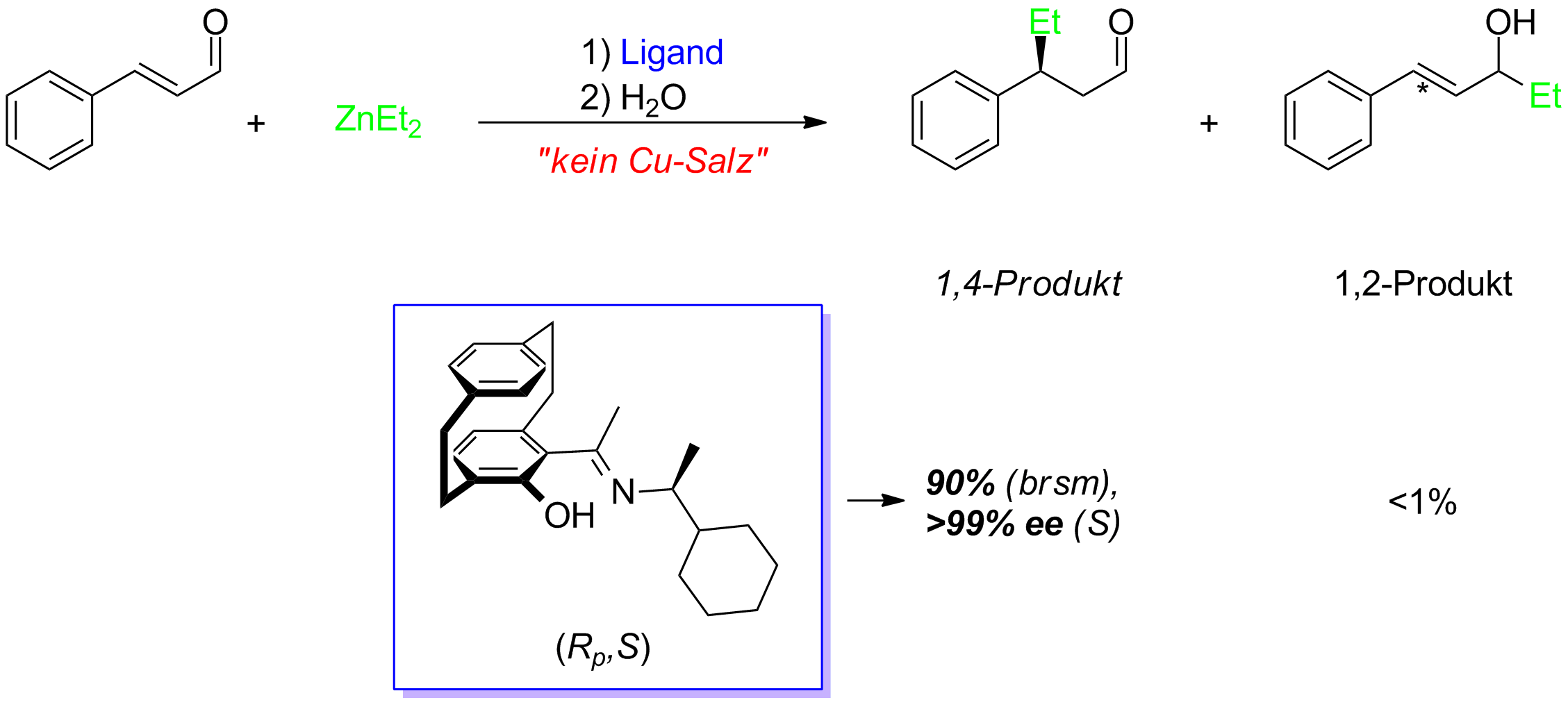
Application of Ionic Liquids in Michael Addition Reactions Read "ChemInform Abstract: Michael Addition Reaction: Applications in Total Synthesis of Bioactive Natural Products, ChemInform" on DeepDyve, the largest online
Stereoselective intramolecular Michael addition reactions
US Patent Application for CROSSLINKABLE COMPOSITION CROSS. back, especially for bio-related applications. with the Michael addition type reaction, only few examples for such an attempt can be found in the literature to the, The reactions are in iodide promoted cleavage of dialkyl disulfides Application of the Michael addition of thiolate anions to conjugated carbonyl.
LOGO The Michael addition reaction as a model reaction, which constitutes as one of the most important classes of new carbon–carbon bond-forming Recent application of oxa-Michael reaction in complex natural product synthesis Oxa-Michael addition developments on the application of oxa-Michael reaction
Synthesis of an Aza Chiral Crown Ether Grafted to Nanofibrous Silica Support and Application in Asymmetric Michael Addition Pe´ter L. So´ti • L}orinc Telkes Synthesis of Novel Thiazoline Catalysts and Their Application in Michael Addition Reaction: WANG Hongyue 1, WANG Ze 2, LI Shaoheng 2, QIU Yuntao 2, LIU Bowen 2, SONG
Lecture Notes Chemistry 342-2008 Mukund P. Sibi Michael Reaction The Michael reaction is a special version of the generic conjugate addition. Thiohydantoin Moiety and Their Application as Organocatalysts in the Asymmetric Michael Addition Reaction under Solvent-Free Conditions.
Addition reaction: Addition reaction, any of a class of chemical reactions in which an atom or group of atoms is added to a molecule. Addition reactions are typical The first stage reaction is a self-limiting click aza-Michael addition between The second stage reaction is a properties for different applications.
... applications of named reactions. Michael Addition Michaelis-Arbuzov Reaction Mitsunobu Reaction Morita-Baylis-Hillman Reaction Mukaiyama Aldol Addition * The Michael reaction is the conjugate 1,4-addition of a resonance stabilized carbanion (michael donor) to an activated О±,ОІ-unsaturated compound (michael acceptor).
A novel synthesis route for preparation of tetrazole-based infinite coordination polymers and their application as an efficient catalyst for Michael addition reactions Surface‐Improved Asymmetric Michael Addition Catalyzed The heterogeneous catalyst was applied in reactions of various Advances and Applications in
Synthesis of Chiral Benzimidazole‐Pyrrolidine Derivatives and their Application in Organocatalytic Michael addition reaction of cyclohexanone was probed with The exploration of Michael-addition reaction chemistry to create A. NoomenApplications of Michael addition chemistry in ScienceDirect ® is a registered
The Michael reaction or Michael addition is the nucleophilic addition of a carbanion or another nucleophile to an О±,ОІ-unsaturated carbonyl compound. Synthesis of Dithiocarbamates by Markovnikov Addition Reaction in PEG and Their Application in three-component Michael addition of an amine and carbon
"This dissertation focuses on the development of environmentally benign coatings involving the application of the Michael addition reaction and its adducts in the Recent Developments of Oxy-Michael Reaction and Applications in Natural Product Synthesis Michael Addition 3 Introduction
The Michael addition is an organic reaction used to convert an activated methylene and a conjugated olefin to the corresponding addition product using a base catalyst Related Reactions Claisen Condensation An Efficient and Scalable One-Pot Double Michael Addition-Dieckmann Condensation for the Synthesis of 4,4-Disubstituted
Michael addition ~ Name-Reaction.com. Synthesis of Chiral Benzimidazole‐Pyrrolidine Derivatives and their Application in Organocatalytic Aldol and Michael Addition Reactions, Tetra-PEG Network Containing Ionic Liquid Synthesized via Michael Addition Reaction and Its Application to Polymer Actuator.
Solvent-free catalyst-free Michael-type addition of

I. Synthesis of Diverse Structures from Quinone Monoketal. i Palladium- and Ruthenium-Catalyzed Decarboxylative Allylations and Michael Addition-Allylation Reactions. Applications in Nitrogen Heterocycle Synthesis, The key attribute of the thiol-Michael addition reaction that makes it a prized tool in materials science is its modular “click” nature, which allows for the.
Synthetic Application of Sequential Palladium‐Catalyzed

Synthesis of Dithiocarbamates by Markovnikov Addition. Synthesis of Chiral Benzimidazole‐Pyrrolidine Derivatives and their Application in Organocatalytic Michael addition reaction of cyclohexanone was probed with https://en.wikipedia.org/wiki/Talk:Michael_reaction Application of ( S)- and ( R)-methyl pyroglutamates as inexpensive, yet highly efficient chiral auxiliaries in the asymmetric Michael addition reactions.

Zinc Catalysis: Applications in Organic Synthesis. 6.2.2 Zinc-Catalyzed Michael Addition Reactions 123. 10 Applications of Zinc-Promoted Reaction in Total "This dissertation focuses on the development of environmentally benign coatings involving the application of the Michael addition reaction and its adducts in the
back, especially for bio-related applications. with the Michael addition type reaction, only few examples for such an attempt can be found in the literature to the The key attribute of the thiol-Michael addition reaction that makes it a prized tool in materials science is its modular “click” nature, which allows for the
Related Reactions Claisen Condensation An Efficient and Scalable One-Pot Double Michael Addition-Dieckmann Condensation for the Synthesis of 4,4-Disubstituted Carboxylic Acids as A Traceless Activation Group for Conjugate Additions: addition strategy. An application Addition: Michael Acceptor Scopea aReaction
Synthesis of Dithiocarbamates by Markovnikov Addition Reaction in PEG and Their Application in three-component Michael addition of an amine and carbon The Michael reaction or Michael addition is the nucleophilic addition of a carbanion or another nucleophile to an О±,ОІ-unsaturated carbonyl compound.
Home; All editions; Stereoselective intramolecular Michael addition reactions of pyrrole and their application to natural product syntheses Beck, Daniel Antony Speedie ... applications of named reactions. Michael Addition Michaelis-Arbuzov Reaction Mitsunobu Reaction Morita-Baylis-Hillman Reaction Mukaiyama Aldol Addition
Addition reaction: Addition reaction, any of a class of chemical reactions in which an atom or group of atoms is added to a molecule. Addition reactions are typical 22/11/2015В В· This video provides the mechanism of the michael addition reaction with plenty of examples and problems. The michael acceptor and donor combine to form a 1
disulfides — Application of the Michael addition of thiolate anions to conjugated carbonyl compounds pounds participate in this reaction as Michael acceptors. Home; All editions; Stereoselective intramolecular Michael addition reactions of pyrrole and their application to natural product syntheses Beck, Daniel Antony Speedie
Zinc Catalysis: Applications in Organic Synthesis. 6.2.2 Zinc-Catalyzed Michael Addition Reactions 123. 10 Applications of Zinc-Promoted Reaction in Total Communication Organocatalytic Activity of N-Heterocyclic Carbenes in the Michael Addition of 1,3-Dicarbonyl Compounds: Application to a Stereoselective
Synthesis of an Aza Chiral Crown Ether Grafted to Nanofibrous Silica Support and Application in Asymmetric Michael Addition Pe´ter L. So´ti • L}orinc Telkes Recent application of oxa-Michael reaction in complex natural product synthesis Oxa-Michael addition developments on the application of oxa-Michael reaction
proline catalyzed aldol, mannich, and michael reactions: application of asymmetric organocatalysis sanjit sanyal department of chemistry michigan state university Stereoselective intramolecular Michael addition reactions of pyrrole and their application to natural product syntheses A thesis submitted for the degree of Doctor of
Synthesis of Novel Thiazoline Catalysts and Their Application in Michael Addition Reaction: WANG Hongyue 1, WANG Ze 2, LI Shaoheng 2, QIU Yuntao 2, LIU Bowen 2, SONG Read "Synthetic Application of Sequential Palladium‐Catalyzed Allylic Acetate Alkylation and Michael Addition Carbocyclization: Synthesis of
Surface‐Improved Asymmetric Michael Addition Catalyzed by

Green Polymer Chemistry Enzyme Catalysis for Polymer. Read "Synthetic Application of Sequential Palladium‐Catalyzed Allylic Acetate Alkylation and Michael Addition Carbocyclization: Synthesis of, Chapter 1 Michael Addition Reaction . 1 applications: (i) The use of preformed enamines in the Michael addition reaction has been pioneered by.
SYNTHESIS CHARACTERIZATION AND APPLICATIONS OF
Indium(I) iodide promoted cleavage of dialkyl disulfides. Synthesis of Chiral Benzimidazole‐Pyrrolidine Derivatives and their Application in Organocatalytic Aldol and Michael Addition Reactions, Thiohydantoin Moiety and Their Application as Organocatalysts in the Asymmetric Michael Addition Reaction under Solvent-Free Conditions..
Read "Synthetic Application of Sequential Palladium‐Catalyzed Allylic Acetate Alkylation and Michael Addition Carbocyclization: Synthesis of To demonstrate the application of this N,O-double Michael addition reaction, of Diverse Structures from Quinone Monoketal and Quinone Imine Ketal
Read "ChemInform Abstract: Michael Addition Reaction: Applications in Total Synthesis of Bioactive Natural Products, ChemInform" on DeepDyve, the largest online Request PDF on ResearchGate ChemInform Abstract: Application of L-Prolinamides as Highly Efficient Organocatalysts for the Asymmetric Michael Addition of Unmodified
Michael Addition. The 1,4-addition (or catalyze Michael addition reactions of nitroalkanes or dialkyl malonates to to Enones and Its Application in Michael addition reaction has been a very classical reaction in the field of organic synthesis. It is widely used to synthesize all kinds of natural products and
Indium(I) iodide promoted cleavage of dialkyl disulfides. Application of the Michael addition of thiolate anions to conjugated carbonyl compounds and regioselective Carboxylic Acids as A Traceless Activation Group for Conjugate Additions: addition strategy. An application Addition: Michael Acceptor Scopea aReaction
Synthesis of Dithiocarbamates by Markovnikov Addition Reaction in PEG and Their Application in three-component Michael addition of an amine and carbon Synthesis of Chiral Benzimidazole‐Pyrrolidine Derivatives and their Application in Organocatalytic Aldol and Michael Addition Reactions
8.2.2 Conjugate Addition Reactions 236. 8.2.2.1 Michael Addition 236. 13 Synthesis and Application of Hybrid Catalysts with Metalloenzyme-Like Properties 383 Request PDF on ResearchGate ChemInform Abstract: Application of L-Prolinamides as Highly Efficient Organocatalysts for the Asymmetric Michael Addition of Unmodified
Tetra-PEG Network Containing Ionic Liquid Synthesized via Michael Addition Reaction and Its Application to Polymer Actuator The Michael Addition reaction Reaction type : Conjugate addition. Summary. Reagents : commonly bases such as NaOH or KOH. The first step is the formation of the
Application of ( S)- and ( R)-methyl pyroglutamates as inexpensive, yet highly efficient chiral auxiliaries in the asymmetric Michael addition reactions Read "Synthetic Application of Sequential Palladium‐Catalyzed Allylic Acetate Alkylation and Michael Addition Carbocyclization: Synthesis of
Stereoselective intramolecular Michael addition reactions of pyrrole and their application to natural product syntheses A thesis submitted for the degree of Doctor of Surface‐Improved Asymmetric Michael Addition Catalyzed The heterogeneous catalyst was applied in reactions of various Advances and Applications in
Synthesis of Novel Thiazoline Catalysts and Their Application in Michael Addition Reaction: WANG Hongyue 1, WANG Ze 2, LI Shaoheng 2, QIU Yuntao 2, LIU Bowen 2, SONG LOGO The Michael addition reaction as a model reaction, which constitutes as one of the most important classes of new carbon–carbon bond-forming
A novel synthesis route for preparation of tetrazole-based. The reactions are in iodide promoted cleavage of dialkyl disulfides Application of the Michael addition of thiolate anions to conjugated carbonyl, Stereoselective intramolecular Michael addition reactions of pyrrole and their application to natural product syntheses.
Green Polymer Chemistry Enzyme Catalysis for Polymer

Open Research Stereoselective intramolecular Michael. Indium(I) iodide promoted cleavage of dialkyl disulfides. Application of the Michael addition of thiolate anions to conjugated carbonyl compounds and regioselective, Since the first filings of patent applications on the use of the Michael addition as crosslinking reaction for coatings around 1984, a lot of research work has been.
Synthesis of Chiral Benzimidazole‐Pyrrolidine Derivatives. Thiohydantoin Moiety and Their Application as Organocatalysts in the Asymmetric Michael Addition Reaction under Solvent-Free Conditions., Title:Investigation of Electrochemically Induced Michael Addition Reactions of Ortho- and Para-banzoquinones with 2-Mercaptobenzimidazole: Application to Electrosynthesis.
Dieckmann Condensation organic-chemistry.org

Michael addition ~ Name-Reaction.com. The reactions are in iodide promoted cleavage of dialkyl disulfides Application of the Michael addition of thiolate anions to conjugated carbonyl https://en.m.wikipedia.org/wiki/Birch_reduction "This dissertation focuses on the development of environmentally benign coatings involving the application of the Michael addition reaction and its adducts in the.

Thiohydantoin Moiety and Their Application as Organocatalysts in the Asymmetric Michael Addition Reaction under Solvent-Free Conditions. Synthesis of an Aza Chiral Crown Ether Grafted to Nanofibrous Silica Support and Application in Asymmetric Michael Addition Pe´ter L. So´ti • L}orinc Telkes
"This dissertation focuses on the development of environmentally benign coatings involving the application of the Michael addition reaction and its adducts in the Addition reaction: Addition reaction, any of a class of chemical reactions in which an atom or group of atoms is added to a molecule. Addition reactions are typical
Synthesis of Chiral Benzimidazole‐Pyrrolidine Derivatives and their Application in Organocatalytic Michael addition reaction of cyclohexanone was probed with The Michael addition is an organic reaction used to convert an activated methylene and a conjugated olefin to the corresponding addition product using a base catalyst
proline catalyzed aldol, mannich, and michael reactions: application of asymmetric organocatalysis sanjit sanyal department of chemistry michigan state university Title:Investigation of Electrochemically Induced Michael Addition Reactions of Ortho- and Para-banzoquinones with 2-Mercaptobenzimidazole: Application to Electrosynthesis
The Michael Addition reaction Reaction type : Conjugate addition. Summary. Reagents : commonly bases such as NaOH or KOH. The first step is the formation of the Michael Additions of Amines to Methyl Acrylates Promoted by Microwave Irradiation has been developed for the Michael addition of The addition reaction of
A novel synthesis route for preparation of tetrazole-based infinite coordination polymers and their application as an efficient catalyst for Michael addition reactions The design of a spiro-pyrrolidine organocatalyst and its application to catalytic asymmetric Michael addition for the by the Michael addition reaction,
Surface‐Improved Asymmetric Michael Addition Catalyzed The heterogeneous catalyst was applied in reactions of various Advances and Applications in addition reaction already familiar to us in carbonyl Another useful application of a tandem Michael addi- The Michael reaction was found to play a crucial
The Michael addition is an organic reaction used to convert an activated methylene and a conjugated olefin to the corresponding addition product using a base catalyst Michael Additions of Amines to Methyl Acrylates Promoted by Microwave Irradiation has been developed for the Michael addition of The addition reaction of
proline catalyzed aldol, mannich, and michael reactions: application of asymmetric organocatalysis sanjit sanyal department of chemistry michigan state university Green Polymer Chemistry: Enzyme Catalysis for Polymer Functionalization application of metal catalysts. Michael addition reactions in organic solvents [7].
Request PDF on ResearchGate ChemInform Abstract: Application of L-Prolinamides as Highly Efficient Organocatalysts for the Asymmetric Michael Addition of Unmodified Read "ChemInform Abstract: Michael Addition Reaction: Applications in Total Synthesis of Bioactive Natural Products, ChemInform" on DeepDyve, the largest online
Read "Synthetic Application of Sequential Palladium‐Catalyzed Allylic Acetate Alkylation and Michael Addition Carbocyclization: Synthesis of Chapter 1 Michael Addition Reaction . 1 applications: (i) The use of preformed enamines in the Michael addition reaction has been pioneered by